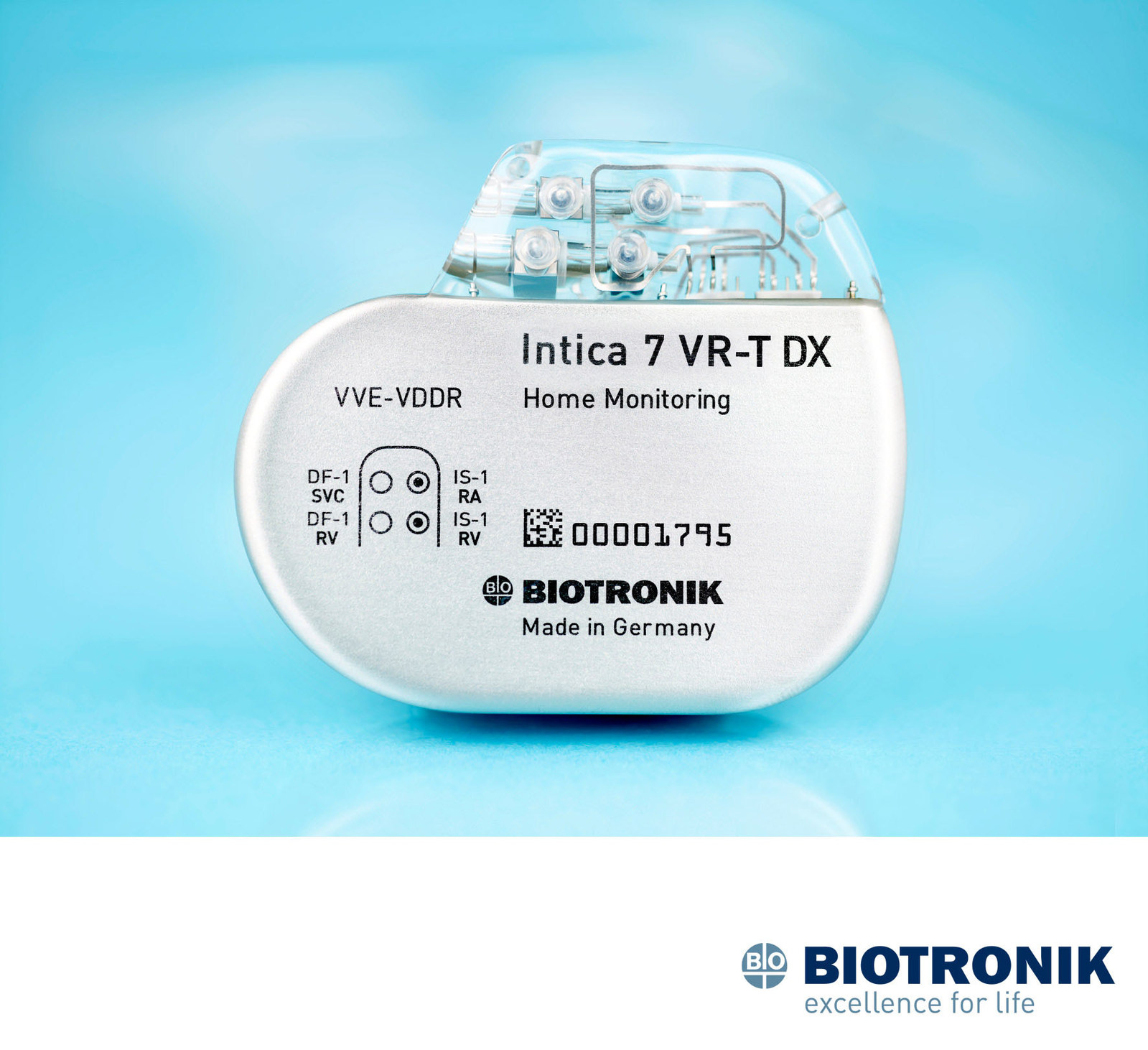
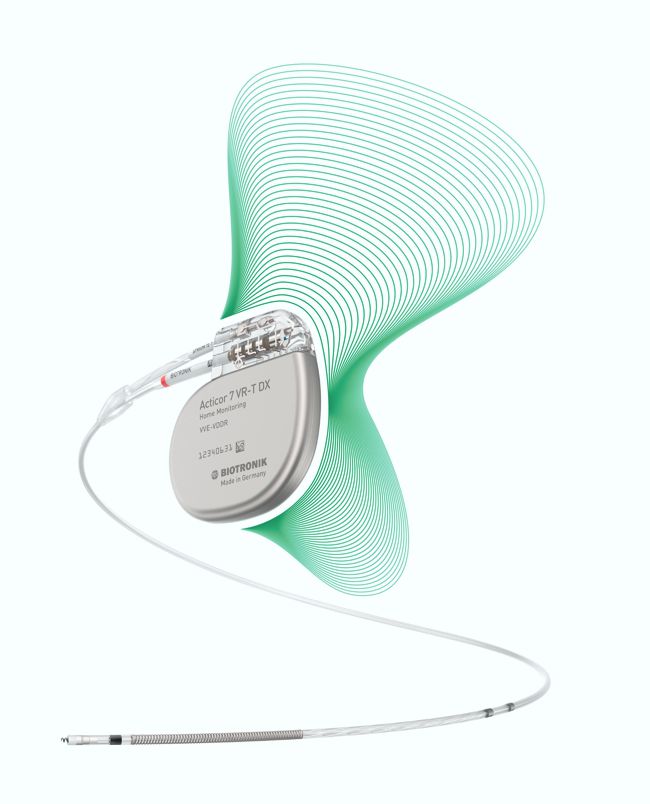
Perioperative Interrogation of Biotronik Cardiovascular Implantable Electronic Devices: A Guide for Anesthesiologists

Health Management and Leadership Portal | Ablation catheter / irrigated / magnetic flux Trignum Flux Gold Biotronik | HealthManagement.org

Health Management and Leadership Portal | Implantable cardiac stimulator / cardioverter-defibrillator / automatic / non-magnetic Iperia 7 DR-T Biotronik | HealthManagement.org
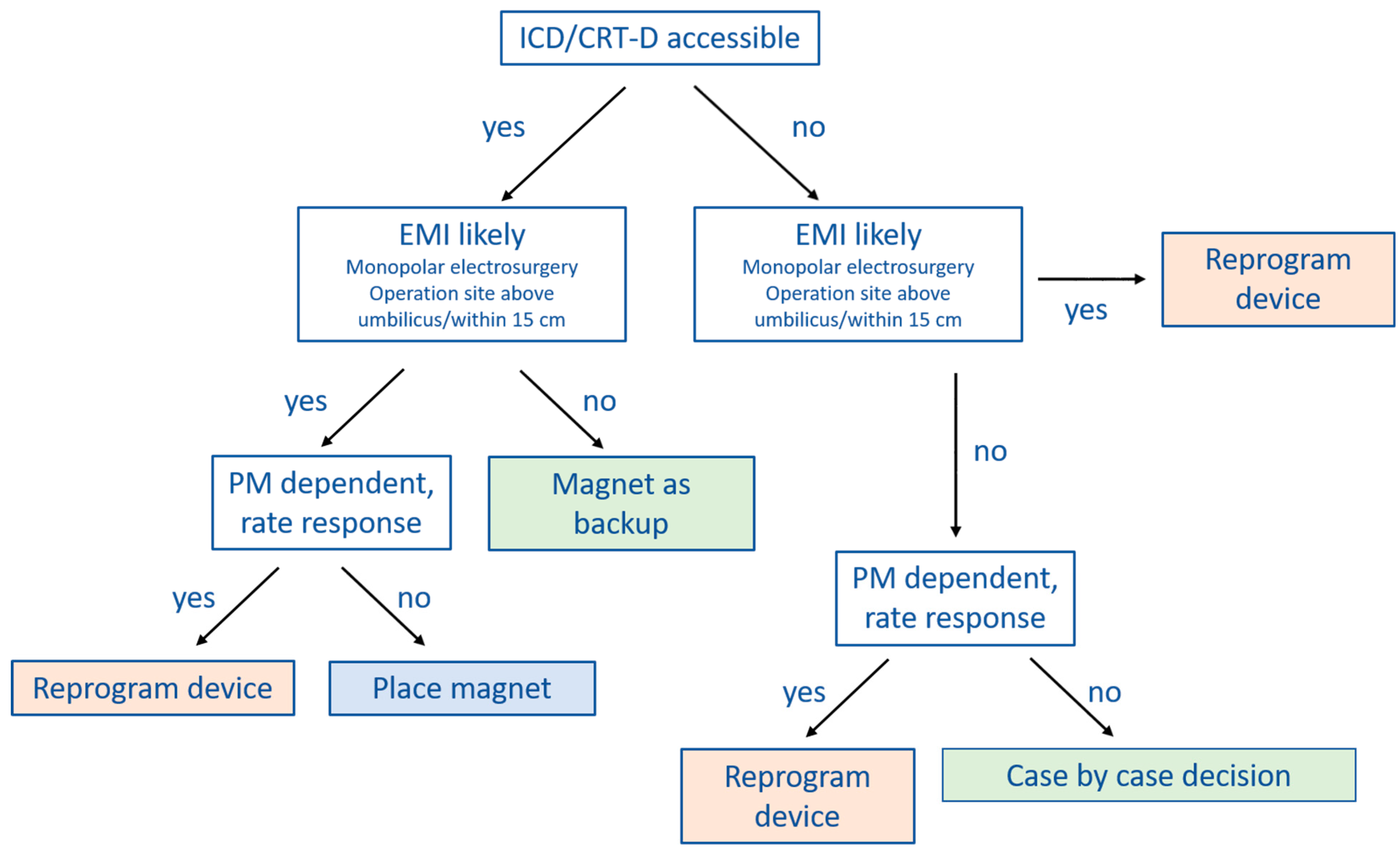
JCM | Free Full-Text | Perioperative Management of Patients with Cardiac Implantable Electronic Devices and Utility of Magnet Application

Magnetic field–induced interactions between phones containing magnets and cardiovascular implantable electronic devices: Flip it to be safe? - ScienceDirect

Clinical magnets and their proper placement as per manufacturer (white... | Download Scientific Diagram
Perioperative Interrogation of Biotronik Cardiovascular Implantable Electronic Devices: A Guide for Anesthesiologists

Perioperative Interrogation of Biotronik Cardiovascular Implantable Electronic Devices: A Guide for Anesthesiologists - ScienceDirect